Section: New Results
Results of a multicenter randomized placebo-controlled clinical trial in prodromal Alzheimer’s disease
Participants : Bruno Dubois, Marie Chupin, Harald Hampel, Simone Lista, Enrica Cavedo, Bernard Croisille, Guy Louis Tisserand, Jacques Touchon, Alain Bonafé, Pierre-Jean Ousset, Amir Ait Ameur, Olivier Rouaud, Frédéric Ricolfi, Alain Viguetto, Florence Pasquier, Christine Delmaire, Mathieu Ceccaldi, Nadine Girard, Carole Dufouil, Stéphane Lehéricy, Isabelle Tonelli, Françoise Duveau, Olivier Colliot, Line Garnero, Marie Sarazin, Didier Dormont [Correspondant] .
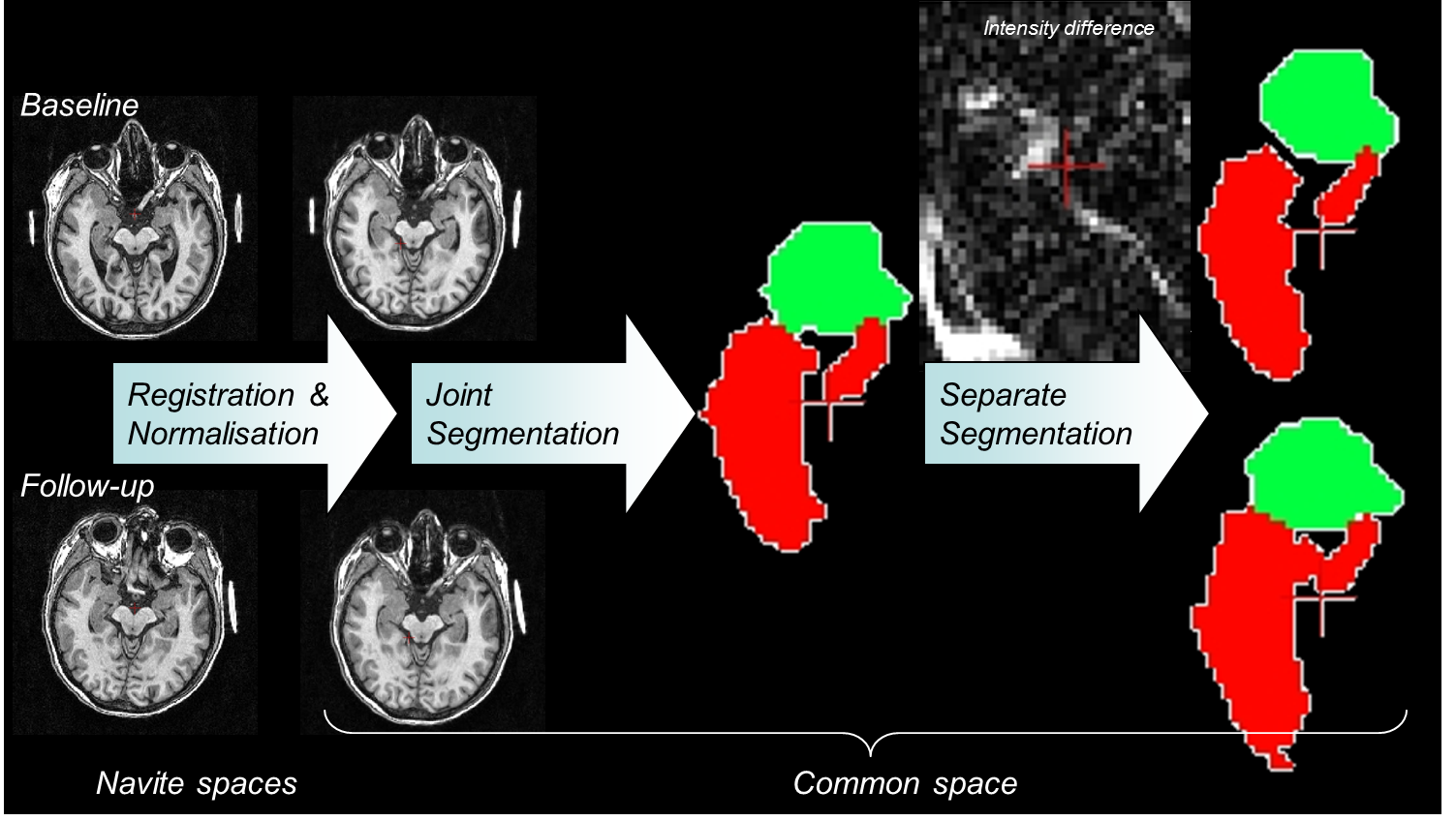
Our team coordinated neuroimage acquisition and analysis of a multicenter randomized placebo-controlled clinical trial aiming to assess the efficacy of donepezil in prodromal Alzheimer’s disease. Subjects underwent two brain magnetic resonance imaging scans (baseline and final visit). The primary efficacy outcome was the annualized percentage change (APC) of total hippocampal volume (left + right) measured by the software (see Section SACHA 6.3 ) developped by our team. Two-hundred and sixteen only subjects were randomized across 28 French expert clinical sites. In the per protocol population (placebo = 92 and donepezil = 82), the donepezil group exhibited a significant reduced rate of hippocampal atrophy (APC= -1.89%) compared with the placebo group (APC= -3.47%), P <.001. There was no significant difference in neuropsychological performance between treatment groups. A 45% reduction of rate of hippocampal atrophy was observed in prodromal AD following 1 year of treatment with donepezil compared with placebo.
This new approach opens interesting perspectives for the evaluation of treatments in neurodegenerative diseases.
More details in [12] .
|