Section: New Results
Modeling
Collective effect in molecular motors assembly
Participants : Matthieu Caruel, Jean-Marc Allain [Ecole Polytechnique] , Lev Truskinovsky [Ecole Polytechnique] .
Skeletal muscles consist of active material capable of producing force. At the microscale, force is the result of complex interactions between two types of proteins named actin and myosin that work coherently in very large assemblies (). The passive mechanical response of skeletal muscles at fast time scales is dominated by long range interactions inducing cooperative behavior without breaking the detailed balance. This leads to such unusual ‘‘material properties’’ as negative equilibrium stiffness and different behavior in force and displacement controlled loading conditions. Our fitting of experimental data suggests that ‘‘muscle material’’ is finely tuned to perform close to a critical point which explains large fluctuations observed in muscles close to the stall force. See paper [28] .
Dimensional reductions of a cardiac model for effective validation and calibration
Participants : Matthieu Caruel, Alexandre Imperiale, Radomir Chabiniok [King's College London] , Philippe Moireau, Dominique Chapelle, Yves Lecarpentier [Institut du Cœur] .
Complex 3D beating heart models are now available, but their complexity makes calibration and validation very difficult tasks. We thus propose a systematic approach of deriving simplified reduced-dimensional models, in “0D”— typically, to represent a cardiac cavity, or several coupled cavities and in “1D”—to model elongated structures such as muscle samples or myocytes. We apply this approach with an earlier-proposed 3D cardiac model designed to capture length-dependence effects in contraction, which we here complement by an additional modeling component devised to represent length-dependent relaxation. We then present experimental data produced with rat papillary muscle samples when varying preload and afterload conditions, and we achieve some detailed validations of the 1D model with these data, including for the length-dependence effects that are accurately captured. Finally, when running simulations of the 0D model pre-calibrated with the 1D model parameters, we obtain pressure–volume indicators of the left ventricle in good agreement with some important features of cardiac physiology, including the so-called Frank–Starling mechanism, the End-Systolic Pressure–Volume Relationship, as well as varying elastance properties. This integrated multi-dimensional modeling approach thus sheds new light on the relations between the phenomena observed at different scales and at the local versus organ levels. See papers [13] , [22] .
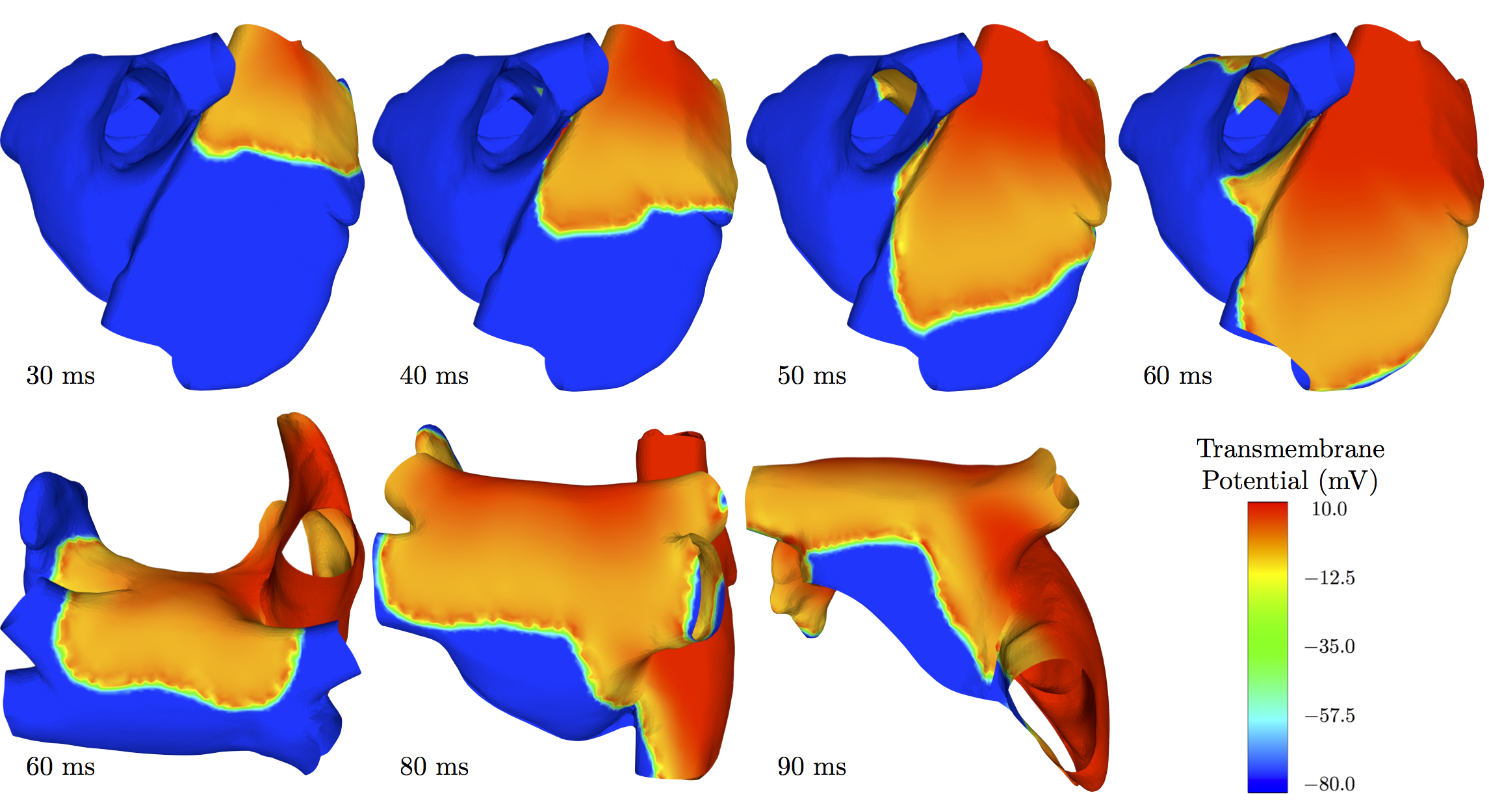
Surface-based electrophysiology modeling and assessment of physiological simulations in atria
Participants : Dominique Chapelle, Annabelle Collin, Jean-Frédéric Gerbeau [Reo Project-Team] , Mélèze Hocini [Institut LIRYC - IHU Bordeaux] , Michel Haïssaguerre [Institut LIRYC - IHU Bordeaux] .
The objective of this work is to assess a previously-proposed surface-based electrophysiology model with detailed atrial simulations. This model – derived and substantiated by mathematical arguments – is specifically designed to address thin structures such as atria, and to take into account strong anisotropy effects related to fiber directions with possibly rapid variations across the wall thickness. The simulation results are in excellent adequacy with previous studies, and confirm the importance of anisotropy effects and variations thereof, see Figure 1 . Furthermore, this surface-based model provides dramatic computational benefits over 3D models with preserved accuracy. See paper [23] .
Strong convergence results in the asymptotic behavior of the 3D-shell model
Participants : Dominique Chapelle, Annabelle Collin.
The objective of this work is to revisit the asymptotic convergence properties – with respect to the thickness parameter – of the earlier-proposed 3D-shell model. This shell model is very attractive for engineering applications, in particular due to the possibility of directly using a general 3D constitutive law in the corresponding finite element formulations. We establish strong convergence results for the 3D-shell model in the two main types of asymptotic regimes, namely, bending- and membrane-dominated behavior. This is an important achievement, as it completely substantiates the asymptotic consistency of the 3D-shell model with 3D linearized isotropic elasticity. See paper [14] .
Mechanical modeling and numerical methods for poromechanics: Applications to cardiac perfusion
Participants : Bruno Burtschell, Dominique Chapelle, Philippe Moireau.
We have previously formulated a rather general modeling framework of poromechanics – formulations that combine solid and fluid components to represent the behavior of a porous medium – to take into account large deformations and rapid fluid flows, see [29] . This allows to consider, in particular, the application of blood perfusion within the cardiac tissue, which – indeed – features these specific complex phenomena, out of the scope of classical poromechanical models. One of our major objectives now, within the PhD of Bruno Burtschell, is to propose and assess some associated relevant numerical schemes, which requires special care regarding both space and time discretizations. In a first stage, in order to ease numerical prototyping and assessments, an axisymmetric reduction of the model has been formulated, and some existing algorithms of fluid-structure interaction have been implemented within this axisymmetric framework (in FreeFEM++). The rationale is that our poromechanics formulations feature some rather deep similarities to so-called Arbitrary-Lagrangian-Eulerian (ALE) fluid-structure formulations, hence the latter are considered as a natural starting point for further extensions.
Personalized modeling for cardiac amyloidosis diagnosis
Participants : Alessandro Felder, Dominique Chapelle, Philippe Moireau, Jean-François Deux [Hôpital Henri Mondor] , Thibault Damy [Hôpital Henri Mondor] .
Cardiac amyloidosis is a condition induced by pathological deposition of amyloid proteins within muscle tissue and nerves, thus severely impairing the cardiac function and often requiring cardiac transplantation as the only available treatment. Our objective here in a first stage is to use our previously developed patient-specific modeling methodologies to analyse some clinical cases – based on actual patient data – to better apprehend the impact of the pathology on biomechanical properties. Further perspectives include the modeling of the protein deposition and associated tissue remodeling in order to predict the disease evolution in a patient-specific context. This work is performed in collaboration with medical doctors from Hôpital Henri Mondor (Créteil).