2023Activity reportProject-TeamCARMEN
RNSR: 201121001J- Research center Inria Centre at the University of Bordeaux
- In partnership with:Université de Bordeaux
- Team name: Modélisation et calculs pour l'électrophysiologie cardiaque
- In collaboration with:Institut de Mathématiques de Bordeaux (IMB)
- Domain:Digital Health, Biology and Earth
- Theme:Modeling and Control for Life Sciences
Keywords
Computer Science and Digital Science
- A6.1.1. Continuous Modeling (PDE, ODE)
- A6.1.2. Stochastic Modeling
- A6.1.4. Multiscale modeling
- A6.2.1. Numerical analysis of PDE and ODE
- A6.2.6. Optimization
- A6.2.7. High performance computing
- A6.2.8. Computational geometry and meshes
- A6.3.1. Inverse problems
- A6.3.2. Data assimilation
- A6.3.3. Data processing
- A6.3.5. Uncertainty Quantification
Other Research Topics and Application Domains
- B1.1.2. Molecular and cellular biology
- B1.1.8. Mathematical biology
- B2.2.1. Cardiovascular and respiratory diseases
- B2.2.6. Neurodegenerative diseases
- B2.4.1. Pharmaco kinetics and dynamics
- B2.4.3. Surgery
- B2.6.1. Brain imaging
- B2.6.2. Cardiac imaging
1 Team members, visitors, external collaborators
Research Scientists
- Jacques Henry [INRIA, Emeritus, HDR]
- Peter Langfield [INRIA, Researcher]
- Michael Leguebe [INRIA, Researcher]
- Nejib Zemzemi [INRIA, Researcher]
Faculty Members
- Yves Coudière [Team leader, UNIV BORDEAUX, Professor Delegation, HDR]
- Mostafa Bendahmane [UNIV BORDEAUX, Associate Professor, HDR]
- Mark Potse [UNIV BORDEAUX, researcher with a CDI]
- Lisl Weynans [UNIV BORDEAUX, Associate Professor, HDR]
Post-Doctoral Fellows
- Andony Arrieula [UNIV BORDEAUX, until Nov 2023]
- Laetitia Mottet [UNIV BORDEAUX]
PhD Students
- Zeina Chehade [UNIV BORDEAUX]
- Narimane Gassa [UNIV BORDEAUX]
- Emma Lagracie [UNIV BORDEAUX]
- Niami Nasr [BORDEAUX INP, ATER, from Oct 2023]
- Niami Nasr [UNIV BORDEAUX, until Sep 2023]
- Valentin Pannetier [UNIV BORDEAUX]
Technical Staff
- Wissam Bouymedj [UNIV BORDEAUX, Engineer, from Apr 2023]
- Corentin Prigent [UNIV BORDEAUX, Engineer]
Interns and Apprentices
- Mohamed Ahmane [INRIA, Intern, from Oct 2023]
- Narjess Ben Abid [ENIT TUNIS, Intern, from Nov 2023]
- Emna Bouaine [INRIA, Intern, from May 2023 until Oct 2023]
- Arthur Jean [UNIV BORDEAUX, Intern, from May 2023 until Aug 2023]
- Mokrane Mouzaoui [INRIA, Intern, from Jun 2023 until Aug 2023]
Administrative Assistant
- Flavie Blondel [INRIA]
Visiting Scientists
- Hamza Ammar [University of Tunis El Manar, from May 2023 until Jun 2023]
- Blanca Rodriguez Lopez [Oxford University]
External Collaborator
- Hassaan Bukhari [University of Zaragoza]
2 Overall objectives
The Carmen team develops and uses models and numerical methods to simulate the electrophysiology of the heart from the molecular to the whole-organ scale, and its relation to measurable signals inside the heart and on the body surface. It aims at:
- improving understanding of normal and pathological cardiac electrophysiology,
- improving the efficiency and accuracy of numerical models,
- exploiting all available electrical signals for diagnosis,
- improving understanding and guidance of ablative treatment of cardiac arrhythmia.
The numerical models developed, analyzed, and used by the team incorporate essentially the gating dynamics of the ion channels in the cardiac cell membranes and the heterogeneities of the cardiac tissue, coupling processes on the cellular scale into macroscopic reaction-diffusion models. The team also work on incorporating any new biological knowledge, at any scale, that helps to understand the mechanisms of arrythmias, their diagnosis or treatment. At the same time we use simpler or reduced models to solve the inverse problems related to non-invasive electrical imaging of the heart.
The fields involved in our research are: ordinary and partial differential equations (ODE & PDE), inverse problems, numerical analysis, high-performance computing, image segmentation, and mesh construction.
A main goal of the team is to contribute to the work packages defined in the project of IHU Liryc, an institute founded in 2011 that focuses on cardiac arrhythmia.
We cooperate with physiologists and cardiologists on several projects. The team is building new models and powerful simulation tools that will help to understand the mechanisms behind cardiac arrhythmias and to establish personalized and optimized treatments. A particular challenge consists in making the simulations reliable and accessible to the medical community.
3 Research program
3.1 Complex models for the propagation of cardiac action potentials
The contraction of the heart is coordinated by a complex electrical activation process which relies on about a million ion channels, pumps, and exchangers of various kinds in the membrane of each cardiac cell. Their interaction results in a periodic change in transmembrane potential called an action potential. Action potentials in the cardiac muscle propagate rapidly from cell to cell, synchronizing the contraction of the entire muscle to achieve an efficient pump function. The spatio-temporal pattern of this propagation is related both to the function of the cellular membrane and to the structural organization of the cells into tissues. Cardiac arrythmias originate from malfunctions in this process. The field of cardiac electrophysiology studies the multiscale organization of the cardiac activation process from the subcellular scale up to the scale of the body. It relates the molecular processes in the cell membranes to the propagation process through the multiscale structure of the tissue and organ, to measurable signals in the heart and to the electrocardiogram, an electrical signal on the torso surface.
Several improvements of current models of the propagation of action potentials are being developed in the Carmen team, based on previous work 39 and on the data available at IHU Liryc:
- Enrichment of the current monodomain and bidomain models 39, 48 by accounting for structural heterogeneities of the tissue at cellular and intermediate scales. Here we focus on multiscale analysis techniques applied to the various high-resolution structural data available at IHU Liryc.
- Coupling of the tissues from the different cardiac compartments and conduction systems. Here, we develop models that couple 1D, 2D and 3D phenomena described by reaction- diffusion PDEs.
These models are essential to improve our understanding of cardiac electrical dysfunction. To this aim, we use high-performance computing techniques in order to numerically explore the complexity of these models.
We use these model codes for applied studies in two important areas of cardiac electrophysiology: atrial fibrillation 41 and sudden-cardiac-death (SCD) syndromes 7, 644. This work is performed in collaboration with several physiologists and clinicians both at IHU Liryc and abroad.
3.2 Simplified models and inverse problems
The medical and clinical exploration of the cardiac electric signals is based on accurate reconstruction of the patterns of propagation of the action potential. The correct detection of these complex patterns by non-invasive electrical imaging techniques has to be developed. This involves solving inverse problems that cannot be addressed with the more complex models. We want both to develop simple and fast models of the propagation of cardiac action potentials and improve the solutions to the reconstruction questions of cardiac electrical imaging techniques.
These questions concern the reconstruction of diverse information, such as cardiac activation maps or, more generally, the whole cardiac electrical activity, from high-density body surface electrocardiograms. It is a possibly powerful diagnosis technique, which success would be considered as a breakthrough. Although widely studied during the last decade, the reconstructed activation maps, for instance, are highly inacurate and have a poor clinical interest. It remains a challenge for the scientific community to understand how body surface signals can better inform on the fine details of arrhythmic mechanisms.
The most usual method consists in solving a Laplace equation on the volume delimited by the body surface and the epicardial surface, for which we contribute by:
- studying in depth the dependance of the inverse problem on inhomogeneities in the torso, conductivity values, the geometry, electrode positions, etc., and
- improving the solution to the inverse problem by using new regularization strategies, factorization of boundary value problems, and the theory of optimal control.
In addition, we have started to explore many alternative approaches including:
- using complete propagation models in the inverse problem, like the bidomain or monodomain equations, for instance in order to localize electrical sources,
- constructing data-based models using e.g. statistical learning techniques, which would accurately represent some families of well-identified pathologies, or allow to combine physics and biology-informed models and clinical data, and
- constructing simpler models of the propagation of the activation front, based on eikonal or level-set equations.
3.3 Numerical techniques
We want our numerical simulations to be efficient, accurate, and reliable with respect to the needs of the medical community. Based on previous work on solving the monodomain and bidomain equations 5, 4, 8, 1, we will focus on:
- high-order numerical techniques with respect to the variables with physiological meaning, like velocity, AP duration and restitution properties and
- efficient, dedicated preconditioning techniques coupled with parallel computing.
Existing simulation tools used in our team rely, among others, on mixtures of explicit and implicit integration methods for ODEs, hybrid MPI-OpenMP parallellization, algebraic multigrid preconditioning, and Krylov solvers. New developments include high-order explicit integration methods and task-based dynamic parallellism.
3.4 Cardiac electrophysiology at the microscopic scale
Traditional numerical models of whole-heart physiology are based on the approximation of a perfect muscle using homogenisation methods. However, due to aging and cardiomyopathies, the cellular structure of the tissue changes. These modifications can give rise to life-threatening arrhythmias, the mechanisms of which we are investigating in collaboration with cardiologists at the IHU Liryc. For this research we are building models that describe the strong heterogeneity of the tissue at the cellular level.
The literature on this type of model is still very limited 54. Existing models are two-dimensional 45 or limited to idealized geometries, and use a linear (purely resistive) behaviour of the gap-juction channels that connect the cells. We propose a three-dimensional approach using realistic cellular geometry (Fig. 1), nonlinear gap-junction behaviour, and a numerical approach that can scale to hundreds of cells while maintaining a sub-micrometer spatial resolution (10 to 100 times smaller than the size of a cardiomyocyte). Following preliminary work in this area by us 35, 34, 33 and by others 54 we proposed a European project with 10 partner institutes and a 5.8M€ budget to develop software that can simulate such models, with micrometer resolution, on the scale of millions of cells, using future exascale supercomputers (microcard.eu). This project runs from April 2021 to October 2024, and involves also the Inria teams CAMUS, STORM and CARDAMOM as well as the Inria-led MMG Consortium.

Image of the microstructure from histology, current, insufficient, representation of microstructural defects, and foreseen geometry to be used in microscopic models.
The cell-by-cell bidomain model presents numerous mathematical and computational challenges. First, mathematically, its unusual formulation providing time dynamics as an ordinary differential equation (ODE) at the cell- to-cell connections and cell-to-extracellular matrix interfaces. Second, the ionic model coupled to the non standard transmission conditions, introduce stiff non linear dynamics. Third, the simulation would performing for a billions of myocytes, that can lead to a significantly large system. In the MICROCARD project, we simulate the micromodel using finite volumes, finite elements and boundary element methods.
3.5 Models and tools for ablative therapies
Today, the most effective way to treat arrhythmias is to ablate selected regions of the cardiac tissue. As the lesions have no particular electric property, this creates conduction blocks that stop the disorganized propagation of action potentials. The ablation procedure consists in placing a catheter in contact with the targeted site and deliver energy into the tissue. The energy can be overheating by radio-frequency current, electroporating electric pulses or temperature drop (cryotherapy). In practice, the choice of the ablation site is done by the clinician based on previous signal measurements and imagery, and is also guided during the procedure with real-time measurement of the electric signal.
Our team works on several subjects related to ablation techniques that may improve the success rate of the treatments:
- accurate computation of electric fields generated by catheters: complex catheter shapes, contact models, tissue heterogeneities;
- models of creation of the lesions, either through temperature rise (radio-frequency) or electroporation; and
- localization tools to help clinicians target the optimal ablation sites, based on both data of previously ablated patients and synthetic data.
4 Application domains
4.1 Scientific context: IHU Liryc
The University Hospital of Bordeaux (CHU de Bordeaux) is equipped with a specialized cardiology hospital, the Hôpital Cardiologique du Haut-Lévêque, where the group of Professor Michel Haïssaguerre has established itself as a global leader in the field of cardiac electrophysiology 43, 42, 37. Their discoveries in the area of atrial fibrillation and sudden cardiac death syndromes are widely acclaimed, and the group is a national and international referral center for treatment of cardiac arrhythmia. Thus the group also sees large numbers of patients with rare cardiac diseases.
In 2011 the group has won the competition for a 40 million euro Investissements d'Avenir grant for the establishment of IHU Liryc, an institute that combines clinical, experimental, and numerical research in the area of cardiac arrhythmia. The institute works in all areas of modern cardiac electrophysiology: atrial arrhythmias, sudden death due to ventricular fibrillation, heart failure related to ventricular dyssynchrony, and metabolic disorders. It is recognized worldwide as one of the most important centers in this area.
The Carmen team was founded as a part of IHU Liryc. We bring applied mathematics and scientific computing closer to experimental and clinical cardiac electrophysiology. In collaboration with experimental and clinical researchers at Liryc we work to enhance fundamental knowledge of the normal and abnormal cardiac electrical activity and of the patterns of the electrocardiogram, and we develop new simulation tools for training, biological, and clinical applications.
4.2 Basic experimental electrophysiology
Our modeling is carried out in coordination with the experimental teams from IHU Liryc. It helps to write new concepts concerning the multiscale organisation of the cardiac action potentials that will serve our understanding in many electrical pathologies. For example, we model the structural heterogeneities at the cellular scale 36 (the MICROCARD project), and at an intermediate scale between the cellular and tissue scales.
At the atrial level, we apply our models to understand the mechanisms of complex arrythmias and the relation with the heterogeneities at the insertion of the pulmonary veins. We will model the heterogeneities specific to the atria, like fibrosis or fatty infiltration 50, 41. These heterogeneities are thought to play a major role in the development of atrial fibrillation.
At the ventricular level, we focus on (1) modeling the complex coupling between the Purkinje network and the ventricles, which is supposed to play a major role in sudden cardiac death, and (2) modeling the heteogeneities related to the complex organization and disorganization of the myocytes and fibroblasts, which is important in the study of infarct scars for instance.
4.3 Clinical electrophysiology
Treatment of cardiac arrhythmia is possible by pharmacological means, by implantation of pacemakers and defibrillators, and by curative ablation of diseased tissue by local heating, freezing or electroporation. In particular the ablative therapies create challenges that can be addressed by numerical means. Cardiologists would like to know, preferably by noninvasive means, where an arrhythmia originates and by what mechanism it is sustained.
We address this issue in the first place using inverse models, which attempt to estimate the cardiac activity from a (high-density) electrocardiogram. A new project aims to perform this estimation on-site in the catheterization laboratory and presenting the results, together with the cardiac anatomy, on the screen that the cardiologist uses to monitor the catheter positions 46, 31.
4.4 Application in Deep Brain Stimulation
Since 2017, we have been working with neurosurgeons from the Bordeaux University Hospital (Pr Cuny and Dr. Engelhardt) on improving the planning technique for deep brain surgery (DBS) for Parkinson's and Essential tremor diseases. DBS is the last resort to treat the symptoms of Parkinson's disease after the drug Levodopa. The surgery consists in placing electrodes in very specific regions of the patient's brain. These regions are unfortunately not visible on the 1.5 Tesla MRI, the most widely available MRI machines in hospitals. The most effective solution to date is to introduce 5 micro-electrodes (MER) to record the activity of neurons in the patient's brain and to prospect by moving the electrodes in order to find the best location. However, this approach renders the surgery very cumbersome because the patient must be awake during the exploration phase. In addition, this phase takes at least 3 hours and mobilizes a neurologist with his staff. The total duration of the operation is between 7 and 8 hours. Many elderly patients do not tolerate this surgery. We have proposed an approach that avoids the prospecting phase and performs surgery under general anesthesia. The idea is to learn on pairs of clinical landmarks and the position of active electrodes in order to predict the optimal position of the DBS from a pre-operative image. This approach simplifies and standardizes surgery planning. We tested several approaches doi:10.3389/fneur.2021.620360. We continue to seek approaches to fully automate the targeting process. We carried out a proof of concept by learning on the clinical database of the Bordeaux University Hospital. The clinical validation of our approach is in progress through a clinical trial which includes patients from the University Hospitals of Bordeaux and Lyon. Pr Cuny has submitted a phase 3 national clinical research hospital project (PHRCN) including 11 CHUs in France which has been accepted by the General Directorate for Care Offers (DGOS). The aim is to compare our new approach to the ones used in the other centers. Inria Bordeaux is a partner in this project and we maintain the OptimDBS software and solve any technical problem related to the compatibility of the MRIs exported by our software and the surgical robots in the different centers.
5 Social and environmental responsibility
5.1 Footprint of research activities
We avoid flying whenever we can.
5.2 Impact of research results
The MICROCARD project, which we coordinate, has energy efficiency as one of its goals. To this end, our partners in the STORM and CAMUS teams are developing methods to increase the time- and energy-efficiency of cardiac simulation codes.
6 Highlights of the year
-
Project EITCardio co-funded by the Région Nouvelle Aquitaine, Inria and the Fondation EDF on electrical impedance tomography for the detection of cardiac arhythmia, also in collaboration with Inria Saclay.
The objective of this project is to develop mathematical methods for solving Electrical Impedance Tomography (EIT) to enhance the resolution of the ECGi (Electrocardiographic Imaging) problem and validate them experimentally. Specifically, the project consists of two parts:
- development of mathematical and numerical methods to solve the inverse problem of EIT in the torso and identify influential parameters for the propagation of the electric field, such as conductivities and organ movements.
- Experimental validation of the ECGi + EIT coupling. This experimental validation will be conducted first within the experimental setup, the torso tank, currently available at Liryc, which allows measurements for ECGi in a controlled environment. Subsequently, it will be conducted as in-vivo experiments, meaning a context closer to clinical reality.
The project involves researchers from the Inria CARMEN team, specializing in modeling the electrical activity of the heart, IHU-Liryc, and LMAP in Pau, experts in scientific computing and uncertainty consideration.
- Project ATLAS-RV funded by the IHU Liryc internal call, led by Peter Langfield, to study typical patterns of repolarization in the human ventricles via in-vivo contact-mapped datasets.
7 New software, platforms, open data
7.1 Evolution of existing software
CEPS.
We added to CEPS the pacemaker models derived for the SimCardioTest project. These models required a complete rewriting of most of CEPS source code, which is now over. We developed new code according the high Software Quality standards, using Inria's sonarQube platform to monitor the issues.
We took advantage of this rewriting to perform a global overhaul of the code in terms of accessibility, be it in the API or the documentation.
7.2 New software
7.2.1 CEPS
-
Name:
Cardiac ElectroPhysiology Simulation
-
Keywords:
Simulation, Health, Mesh, Cardiac, 3D, Cardiac Electrophysiology
-
Scientific Description:
As compared to other existing softwares, CEPS aims at providing a more general framework of integration for new methods or models and a better efficiency in parallel. CEPS is designed to run on massively parallel architectures, and to make use of state-of-the-art and well known computing libraries to achieve realistic and complex heart simulations. CEPS also includes software engineering and validation tools.
-
Functional Description:
CEPS is a modular high-performance computing software for performing numerical simulations in cardiac electrophysiology. It is based on modules : - management of geometries represented by meshes in 3D, 2D or 1D (volumes, surfaces, trees), - model simulation of cellular electrophysiology, - calculating the tissue propagation of the action potentials in the cardiac geometries, - calculation of extracardiac potentials, - time approximation methods in order 2, 3 and 4 specific to electrocardiography.
- URL:
-
Contact:
Ceps Dev Team
-
Participants:
Yves Coudière, Michael Leguebe, Valentin Pannetier, Pierre-elliott Bécue, Florian Caro, Andjela Davidovic, Charlie Douanla Lontsi, Marc Fuentes, Mehdi Juhoor, Pauline Migerditichan, Nejib Zemzemi
-
Partners:
Université de Bordeaux, Fondation Bordeaux Université, CHU de Bordeaux, Inria
7.2.2 OptimDBS
-
Name:
Optimizing the Deep Brain Stimulation
-
Keywords:
Image analysis, Deep brain stimulation, Statistical learning
-
Functional Description:
Targeting software for deep brain stimulation
- URL:
-
Contact:
Nejib Zemzemi
-
Participants:
Nejib Zemzemi, Louise-Amelie Schmitt, Emmanuel Cuny, Julien Engelhardt
-
Partner:
CHU de Bordeaux
7.2.3 MUSIC - Carmen plugins
-
Name:
Carmen plugins for multi-modality imaging in Cardiology
-
Keywords:
Image segmentation, Mesh generation, Image filter, Numerical simulations, Cardiac Electrophysiology, Inverse problem, Finite element modelling, Visualization, 3D interaction, Registration
-
Scientific Description:
Carmen plugins is a collection of toolboxes and pipelines allowing the following functionalities: - Segmenting and filtering the heart, the surrounding organs and the body surface. - Generate/optimize surface and volume of computational meshes for the segmented organs. - Generate fibers orientations for Atria and Ventricles. - Interactively annotate meshes. - Read map and visualize electrical information collected from medical devices. - Numerical simulation of the forward problem using finite elements method. - Method of fundamental solutions for solving the ECGI inverse problem combined with the regularization methods including CRESO,Zero Crossing GCV, RGCV, ADPC and Ucurve. - Landmark based mesh registration. - ECGI pipeline: including segmentation, mesh generation, identification of the vest electrodes.
-
Functional Description:
Carmen plugins is a collection of toolboxes and pipelines allowing the following functionalities: - Segmenting and filtering the heart, the surrounding organs and the body surface. - Generate/optimize surface and volume of computational meshes for the segmented organs. - Generate fibers orientations for Atria and Ventricles. - Interactively annotate meshes. - Read map and visualize electrical information collected from medical devices. - Numerical simulation of the forward problem using finite elements method. - Method of fundamental solutions for solving the ECGI inverse problem combined with the regularization methods including CRESO,Zero Crossing GCV, RGCV, ADPC and Ucurve. - Landmark based mesh registration. - ECGI pipeline: including segmentation, mesh generation, identification of the vest electrodes.
- Publications:
-
Contact:
Nejib Zemzemi
-
Participants:
Hubert Cochet, Florent Collot, Mathilde Merle, Maxime Sermesant, Julien Castelneau, Mehdi Juhoor, Pauline Migerditichan, Nejib Zemzemi, Yves Coudière
-
Partners:
Université de Bordeaux, IHU - LIRYC
Participants: Mark Potse, Andony Arrieula, Laetitia Mottet, Corentin Prigent, Wissam Bouymedj, Yves Coudière.
7.3 New platforms
OpenCARP
OpenCARP is an open cardiac electrophysiology simulator for in-silico experiments. Its source code is public and the software is freely available for academic purposes. OpenCARP is easy to use and offers single cell as well as multiscale simulations from ion channel to organ level. Additionally, openCARP includes a wide variety of functions for pre- and post-processing of data as well as visualization. The python-based CARPutils framework enables the user to develop and share simulation pipelines, i.e. automating in-silico experiments including all modeling/simulation steps.
KIT and the company Numericor, the main creators of openCARP, with the University of Graz, are partners of the MICROCARD project. The openCARP simulator is developed within the MICROCARD project targeting the creation of a microscopiv cardiac model.
We have been contributing by managing the MICROCARD consortium and its contributions to openCARP, and by adapting the partitionning scheme (based on parMETIS) to the context of the microscopic model.
Mmg
Mmg is an open source software suite for simplicial remeshing and an open source Consortium, which participates in the MICROCARD project.
In this context we have been active developers of the Mmg software. Laetitia Mottet (postdoc) works on ParMmg, the parallel version of Mmg, where she implemented level-set discretization and is working on the robustification of the software. Corentin Prigent (engineer) works on the serial code mmg3d for 1) its robustification so that it can deal with the extremely complicated topology of the meshes created using segmented data provided by partners in the MICROCARD project, and 2) for the purposes of the MICROCARD project, Mark Potse uses mmg3d for the challenging task of creating artificial models of cardiac muscle fibers (Figure 1C).
In the MICROCARD project, tetrahedral meshes are created using both segmented data and synthetic models. Several improvements made this year in mmg now allow us to 1) use these segmented data to create valid meshes with much less elements than in the initial data, 2) improve the synthetic generation of cardiac tissue.
CEMPACK
CEMPACK is a collection of software that was previously archived in different places. It includes the high-performance simulation code Propag and a suite of software to create geometric models, prepare inputs for Propag, and analyse its outputs. In 2017 the code was collected in an archive on Inria's GitLab platform. The main components of CEMPACK are the following.
-
Propag-5.1
Applied modeling studies performed by the Carmen team in collaboration with IHU Liryc and foreign partners 751, 41, 40, 38 rely on high-performance computations on the national supercomputers Irene, Zay, and Adastra. The Propag-5 code is optimized for these systems. It is the result of a decades-long development first at the Université de Montréal in Canada, then at Maastricht University in the Netherlands, and finally at the Institute of Computational Science of the Università della Svizzera italiana in Lugano, Switzerland. Since 2016 most of the development on Propag has been done by M. Potse at the Carmen team 53. The code scales excellently to large core counts 52 and, as it is controlled completely with command-line flags and configuration files, it can be used by non-programmers. It also features:
- a plugin system for membrane models,
- a completely parallel workflow, including the initial anatomy input and mesh partitioning, which allows it to work with meshes of more than nodes,
- a flexible output scheme allowing hundreds of different state variables and transient variables to be output to file, when desired, using any spatial and temporal subsampling,
- a configurable, LUSTRE-aware parallel output system in which groups of processes write HDF5/netCDF files, and
- CWEB documentation of the entire code base.
The code has been stable and reliable for many years. It can be considered the workhorse for our HPC work until CEPS takes over.
-
Gepetto
The Gepetto suite transforms a surface mesh of the heart into a set of (semi-)structured meshes for use by the Propag software or others. It creates the different fiber orientations in the model, including the transmurally rotating ventricular fibers and the various bundle structures in the atria (figure 2), and creates layers with possibly different electrophysiological properties across the wall. A practically important function is that it automatically builds the matching heart and torso meshes that Propag uses to simulate potentials in the torso (at a resolution of 1 mm) after projecting simulation results from the heart model (at 0.1 to 0.2 mm) on the coarser torso mesh 49. Like Propag, the Gepetto software results from a long-term development that started in Montreal, Canada, around 2002. The code for atrial fiber structure was developed by our team.
-
Blender plugins
Blender is a free software package for the production of 3-D models, renderings, and animations, comparable to commercial software such as Cinema4D. CEMPACK includes a set of plugins for Blender that facilitate the production of anatomical models and the visualization of measured and simulated data. It uses the MMG remeshing library, which is developed by the CARDAMOM team at Inria Bordeaux. Currently our segmentation work is mostly done with the MUSICardio software, but we still use Blender for finishing touches and high-quality visualization.
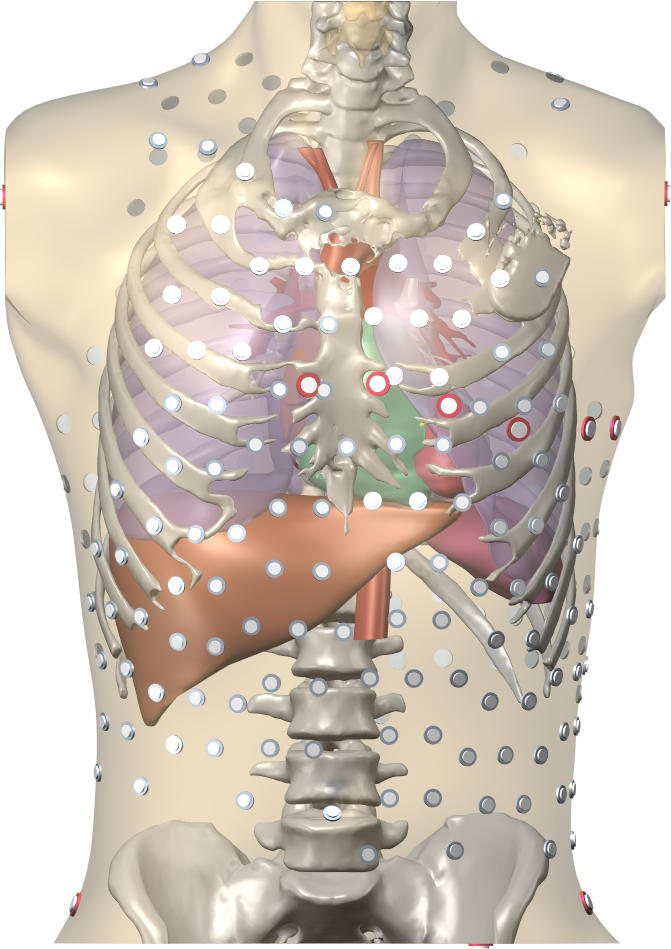
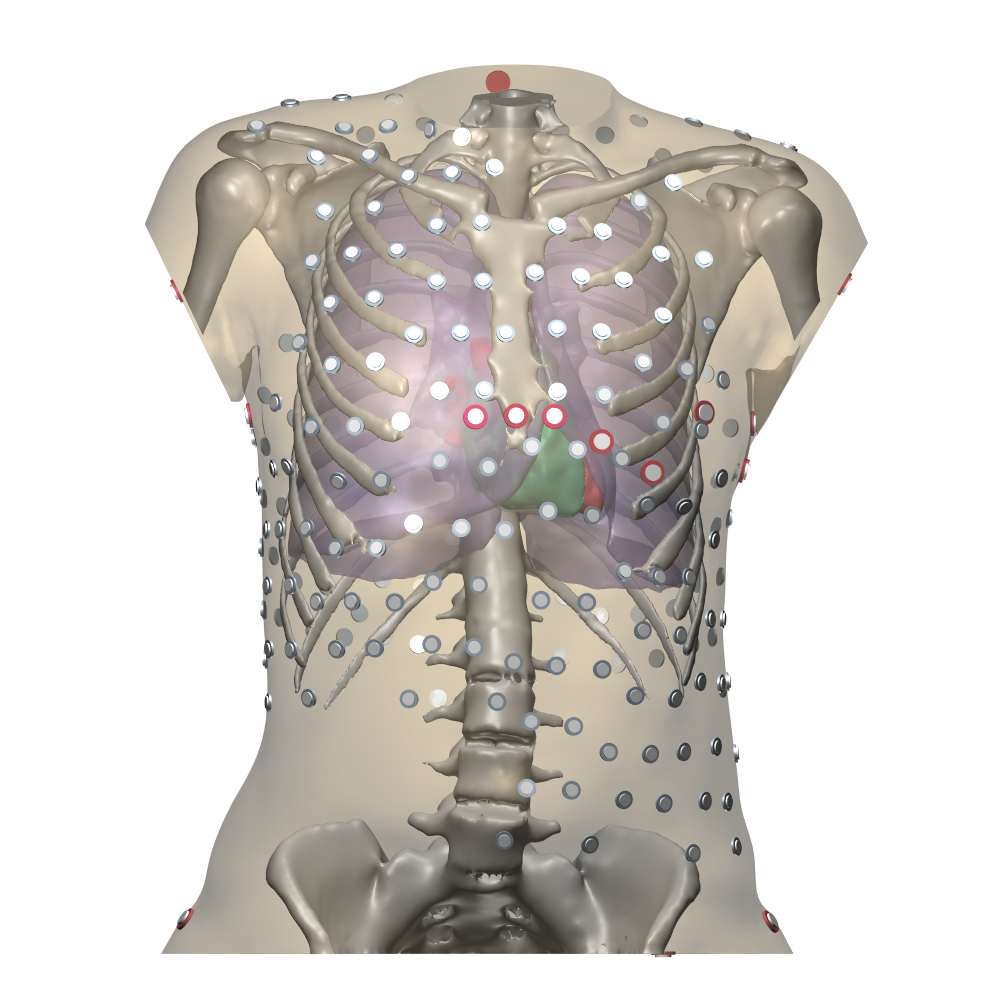
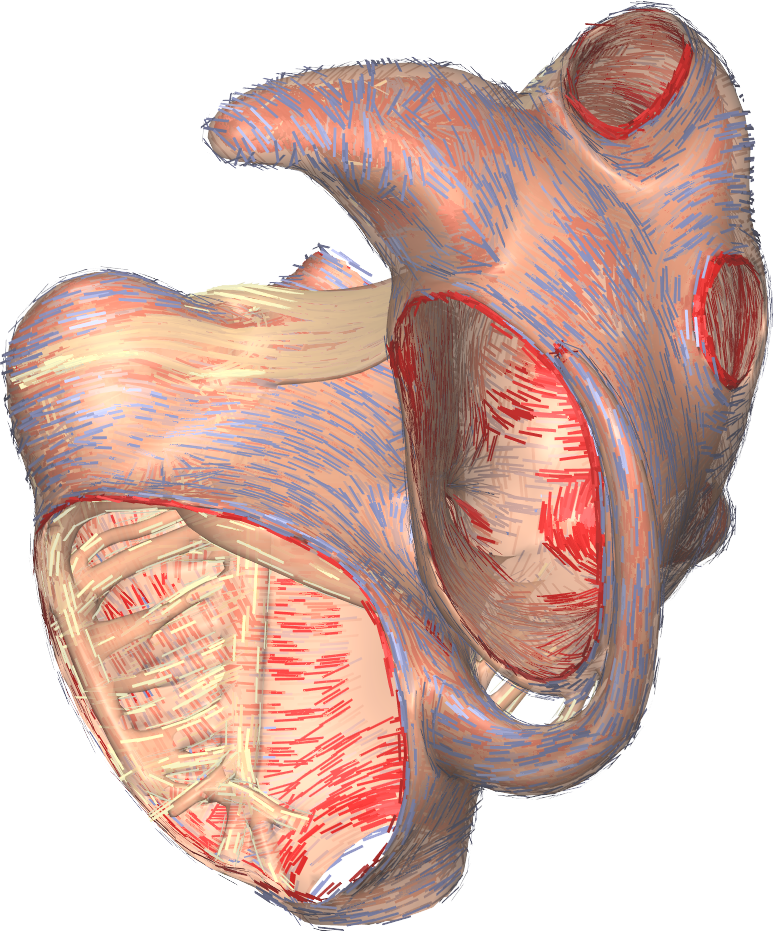
An image of a torso model including the heart, the ribs, the lungs, the livers, and the location of torso electrodes, and a detailed image of an atrial anatomical model including fiber directions.
Participants: Mostafa Bendahmane, Yves Coudière, Jacques Henry, Peter Langfield, Michael Leguèbe, Mark Potse, Lisl Weynans, Nejib Zemzemi.
8 New results
8.1 Numerical analysis and development of numerical methods
- Isochrone computation There has been ongoing development of a numerical continuation-based method to compute higher-dimensional isochrones. The method had been previously applied to the Hodgkin-Huxley model. During this year, a region of extremely low sensitivity of phase response to stimuli was identified, which is a property supportive of synchroniziation between cells. This region was studied and we have hypothesized an underlying mechanism that gives rise to it. These results form a manuscript that is currently in preparation, which will be submitted within the first few months of this year. We also started to use the method to study isochrones in more cardiac-related models, namely, the Beeler-Reuter model and a model of a sinoatrial node cell.
- Immersed boundary method for cardiac EIT We proposed an immersed boundary scheme for the numerical resolution of the Complete Electrode Model in Electrical Impedance Tomography, that we use as a main ingredient in the resolution of inverse problems in medical imaging. Such method allows to use a Cartesian mesh without accurate discretization of the boundary, which is useful in situations where the boundary is complicated and/or changing. We proved the convergence of the method 13.
- Immersed boundary method for some wave equations Besides the issue of cardiac electrophysiology, we also studied numerical schemes to study waves interacting with partially immersed objects allowed to move freely in the vertical direction, and in a regime in which the propagation of the waves is described by the one dimensional Boussinesq-Abbott system 23. The problem can be reduced to a transmission problem for this Boussinesq system, in which the transmission conditions between the components of the domain at the left and at the right of the object are determined through the resolution of coupled forced ODEs in time satisfied by the vertical displacement of the object and the average discharge in the portion of the fluid located under the object. We propose a new extended formulation in which these ODEs are complemented by two other forced ODEs satisfied by the trace of the surface elevation at the contact points. The interest of this new extended formulation is that the forcing terms are easy to compute numerically and that the surface elevation at the contact points is furnished for free. Based on this formulation, we propose a second order scheme that involves a generalization of the MacCormack scheme with nonlocal flux and a source term, which is coupled to a second order Heun scheme for the ODEs. In order to validate this scheme, several explicit solutions for this wave-structure interaction problem are derived and can serve as benchmark for future codes. As a byproduct, our method provides a second order scheme for the generation of waves at the entrance of the numerical domain for the Boussinesq-Abbott system.
8.2 Modeling and inverse problems
- Modeling and in-silico trial for pacemakers We pursued our work within the H2020 SimCardioTest European project, by using both 0D and 3D models of cardiac stimulation by a pacemaker, which were derived in collaboration with Microport CRM, manufacturer. The models couple a model of the pacemaker circuitry with cardiac tissue through boundary conditions which model the polarization of the electrodes. First results were presented at FIMH 2023 18. The implementation of the 3D model was made following high quality standards in order to match the Verification and Validation standards of the V&V40 document 32, which gives a list a software requirements for the Verification aspects. We are currently using data from bench experiments of optical mapping to comply with the Validation items. In particular, we are calibrating parameters of the ionic models using measured activation maps. This verification and validation process following the V& V40 guidelines was presented at the RICAM workshop on Cardiovascular Modeling and simulation in 2023 20. Finally, we designed a first in-silico clinical trial, that is described in a deliverable of the European project (still not published). the question of interest for this trial concerns the changes of the electrical capture properties of a pacemaker bradychardia lead with respect to some design parameters, in the context where fibrosis develops at the tip lf the lead.
- The patchwork method for ECGi We propose a novel “patchwork method” (PM) 10, which combines two classical numerical approaches for ECGI: the method of fundamental solutions (MFS) and the finite-element method (FEM). We assume that the method with the smallest residual in the predicted torso potentials provides the most accurate solution. These residuals were computed using the boundaryelement method, and projected onto the heart surface using an orthogonal projection. The PM then selects for each heart node and each time step the method whose estimated reconstruction error is smallest. The performance of the PM was compared to both FEM and MFS alone using simulated ectopic and normal ventricular activation beats. Results: Cardiac potentials and activation maps obtained with the PM (CC=0.63±0.004 and 0.61±0.05 respectively) were more accurate than MFS (CC=0.61± 0.005 and 0.48±0.05 respectively) or FEM (CC=0.58±0.009 and 0.51±0.02 respectively). The PM also succeeded in locating all epicardial activation breakthrough sites, whereas the individual numerical methods usually missed one. Moreover, the PM outperformed the other methods in the presence of Gaussian measurement noise added to the torso surface potential distribution, showing its robustness and stability with respect to noise. Conclusions: The PM overcomes some of the limitations of classical numerical methods, improving the accuracy of mapping important features of activation during sinus rhythm and paced beats.
- Electrical Impedance Tomography (EIT) We applied the immersed boundary scheme evocated hereabove to the resolution of the inverse problem of Electrical Impedance Tomography. We performed reconstructions of conductivites, contact impedances on electrodes and shape of the considered domain of interest 13.
- A model for cardiac electroporation In the Dielectric project in collaboration with Inria Team MONC and IHU Liryc, we derived formally a bidomain model at the tissue scale that takes into account the specificity of electroporation, which involves different time scales than the classical bidomain model. This model was presented at the 2023 RICAM workshop 26.
- Comparison of different formulations for ECG computations We compared and numerically studied two formulations that map the body surface potential maps from the transmembrane voltage. Those formulations naturally emerge from the bidomain model. We determined which formulation is the most relevant to use in an inverse resolution perpective. We developped a 2D finite volume bidomain code, and compared simplified models with the bidomain 21.
- Impact of cardiac motion on ECGi solutions We conducted an investigation into the impact of cardiac motion on both inverse and forward simulations, as well as the localization of sources from ECGI outcomes. This study relied on clinical patient imaging data, generously provided by Simula Research Laboratory, as part of a research visit conducted during the summer of 2023.
- Numerical Investigation of Methods Used In Commercial Clinical Devices for Solving the ECGI Inverse Problem In this work, we assess two ECGI algorithms used in commercial ECGI systems to solve the inverse problem; the Method of Fundamental Solutions (MFS) and the Equivalent Single Layer (ESL). We quantify the performance of these two methods in conjunction with two different activation maps to estimate the activation times and earliest activation sites (ADM et TID). Results for clinical cases, indicate that ESL combined with ADM resulted in better localization accuracy and, in contrast to simulated data, TID and ADM based earliest activation sites can be very different. In fact, TID based activation maps is less robust to noisy and clinical data. This is mainly due to the fact that TID calculates activation times for each cardiac node separately (thepoint of maximum negative slope) without a spatial coherence. Consequently, in noisy data, it frequently provides patchy patterns in the obtained maps. However, activation direction mapping provides better results for clinical data despitethe fact that it tends to over-smooth the spread of activation, which can bemisleading in the excitation origin localization. The post-processing methods and in particular the activation times calculation have a significant impact on the computation of the Activation map and concequently on PVC localization. 24
- Inter-operator segmentation variability induces high premature ventricular contractions localization uncertainty at the heart base Our main purpose is to evaluate the effect of the inter-operator segmentation variability on the PVC localization. Eight different cardiac segmentations from the same single subject CT-scans were performed by researchers within the consortium for Electrocardiographic Imaging. For all generated meshes, eight ventricular stimulation protocols were used; left and right ventricular free walls (LV, RV), apex, left and right ventricular outflow tract (LVOT, RVOT), septum, and two locations at the left and right heart base (LVB, RVB). BSPs were generated using computational models. We designed two test cases: with and without segmentation uncertainty. We found that the high uncertainty is due to the variability of segmentations at the base of the heart. These findings suggest that uncertainty in cardiac segmentation can have a significant impact on ECGI and its interpretability in clinical applications; therefore, careful segmentation is strongly recommended, especially at the base of the heart. 27
8.3 Clinical applications
- Artificial Intelligence for Detection of Ventricular Oversensing Machine Learning Approaches for Noise Detection Within Non-Sustained Ventricular Tachycardia Episodes Remotely Transmitted by Pacemakers and Implantable Cardioverter Defibrillators Pacemakers (PMs) and implantable cardioverter defibrillators (ICDs) increasingly automatically record and remotely transmit non-sustained ventricular tachycardia (NSVT) episodes which may reveal ventricular oversensing. We aimed at developing and validating a machine learning algorithm which accurately classifies NSVT episodes transmitted by PMs and ICDs in order to lighten healthcare workload burden and improve patient safety. PMs or ICDs (Boston Scientific) from four French hospitals with transmitted NSVT episode were split into three subgroups: training set, validation set, and test set. Each NSVT episode was labelled as either physiological or non-physiological. Four machine learning algorithms (2DTF-CNN, 2D-DenseNet, 2DTF-VGG, and 1D-AgResNet) were developed using a training and validation dataset. Accuracies of the classifiers were compared with an analysis of the remote monitoring team of the Bordeaux University Hospital using F2 scores (favoring sensitivity over predictive positive value) using an independent test set. We used data collected from 807 devices that transmitted 10.471 NSVT recordings (82% ICD, 18% PM), of which 87 devices (10.8%) transmitted 544 NSVT recordings with non-physiological signals. The classification by the remote monitoring team resulted in an F2 score of 0,932 (sensitivity of 95%, specificity of 99%). The four machine learning algorithms showed high and comparable F2 scores (2DTF-CNN: 0,914, 2D-DenseNet: 0,906, 2DTF-VGG: 0,863, 1D-AgResNet: 0,791) and only 1D-AgResNet had significantly different labeling as compared with the remote monitoring team. Our conclusion is that Machine learning algorithms were accurate in detecting non-physiological signals within EGMs transmitted by pacemaker and ICDs. We think that an artificial intelligence approach may render remote monitoring less resourceful and improve patient safety. 15
- Comparison of two segmentation software tools for deep brain stimulation of the subthalamic and ventro-intermedius nuclei Deep brain stimulation (DBS) relies on precise targeting of key structures such as the subthalamic nucleus (STN) for Parkinson's disease (PD) and the ventro-intermedius nucleus of the thalamus (Vim) for essential tremor (ET). Segmentation software, such as GuideXT© de Boston Scientific and Suretune©, de Medtronic commercially available for atlas-based identification of deep brain structures. However, no study has compared the concordance of the segmentation results between the two software. We retrospectively compared the concordance of segmentation of GuideXT© and Suretune© software by comparing the position of the segmented key structures with clinically predicted targets obtained using the newly developed RebrAIn© software as a reference. We targeted the STN in 44 MRI from PD patients (88 hemispheres) and the Vim in 31 MRI from ET patients (62 hemispheres) who were elected for DBS. In 22 STN targeting (25%), the target positioning was not correlating between GuideXT© and Suretune©. Regarding the Vim, targets were located in the segmented Vim in 37%, the posterior subthalamic area (PSA) in 60%, and the STN in 3% of the cases using GuideXT©; the proportions were 34%, 60%, and 6%, respectively, using Suretune©. The mean distance from the centre of the RebrAIn© targeting to the segmented Vim by Suretune© was closer (0.64 mm) than with GuideXT© (0.96 mm; p = 0.0004). 12
8.4 High performance computing and Microscopic models
- A population based study of the effect of ion concentrations on the ECG We have used a population of models to investigate a problem faced by researchers who are trying to infer blood metabolyte concentrations from the ECG. There is in most subjects a clear relation between ECG features and concentrations of sodium and potassium, but these relations differ strongly between subjects. We have shown that inter-subject differences in the heterogeneity of intrinsic cardiac cell properties are a possible explanation for these differences 11.
- Computational methods for the microscopic EMI model The MICROCARD project has developed numerical methods to simulate cardiac electrical activity on cell-by-cell (EMI) models: 1) efficient higher-order time stepping achieved by combining low-overhead spatial adaptivity on the algebraic level with progressive spectral deferred correction methods, and 2) substructuring domain decomposition preconditioners tailored to address the complexities of our heterogeneous problem structures. These methods have been tested using artificial 3D tissue geometries of upto a few hundreds of cells that we generated 16, 19.
- Mesh of a 1 cubic millimeter of cardiac tissue In addition, thanks to improvements in the Mmg code and in mesh generation software we were able to build such meshes of cardiac tissue structure up to 1 cubic millimeter (7 thousand cells, 270 million tetrahedra), a larger volume than can be simulated yet, without resorting to mesh replication. An HPC-capable version of the simulation code is being developed which should be able to handle much larger cases.
- Mesh partitions that respect the geometry of cardiac cells We have also started to work on partitioning these meshes, which requires not only to balance the partitions in accordance with the target machine, but also to keep the cardiac cells intact within each partition. A first prototype was written using the Metis library, and aims at being transferred to the OpenCARP software platoform.
- Finite volume methods for the microscopic EMI model We pursued our work 17 within the MICROCARD European project. The Two-point flux approximation (TPFA) finite volume (FV) scheme was generalized naturally to the extracellular-membrane-intracellular (EMI) model on an admissibile orhtogonal mesh. Under strong regularity assumptions, an error estimates was derrived on the electrical potentials and the transmembrne voltages as well in the case of single myocyte. We believe that extending the proof to the case of many cardiac cells is straightforwad, and the convergence analysis might be easily deduced from the existence proof in 33, even with minimal regularity of the data. In fact, the ODEs from the non-standard transimission conditions on the cell membrane, are coupled with large sets of nonlinear equations that require time-integration specific to the EMI. Indeed, The FV method simplifies the implementation of this coupling by introducing the a Dirichlet to Neuman operator to rewrite the problem as an evolution one on the membrane only. We plan to implement the scheme and compare it to finite elements and a boundary elements discretizations on simple 2D test cases. Afterwards, we aim to generalize the scheme to a FV-like method that is more robust with respect to the more general meshes.
9 Bilateral contracts and grants with industry
Participants: Nejib Zemzemi.
9.1 Bilateral Grants with Industry
- RebrAIn and Inria contracted an agreement allowing Nejib Zemzemi to pass half of his time working at RebrAIn. The startup RebrAIn has been co-founded by N. Zemzemi and E. Cuny.
- Lisl Weynans obtained a grant from Fondation EDF to fund a research project about Electrical Impedance Tomography.
10 Partnerships and cooperations
10.1 International initiatives
10.1.1 Associate Teams in the framework of an Inria International Lab or in the framework of an Inria International Program
SPICY
-
Title:
Stochastic forward and inverse Problems In Cardiac electrophysiologY
-
Duration:
2021 ->
-
Coordinator:
Mourad Bellasoued (mourad.bellassoued@enit.utm.tn)
-
Partners:
- Université de Tunis El Manar Tunis (Tunisie)
-
Inria contact:
Mostafa Bendahmane
-
Summary:
The electrocardiography imaging inverse problem is frequently solved using the deterministic quasi-static models. These models don't take into account the heart dynamic in time, channel noise and external random perturbations acting in the torso. Recent numerical studies in the direct problem have shown that such randomness cannot be suppressed. Occasionally deterministic equations give qualitatively incorrect results. Therefore, it is important to quantify the nature of the noise and choose an appropriate model incorporating randomness. In our project, we study the inverse problem constrained by the stochastic monodomain or bidomain equations in electrocardiology. The state equations consist in a coupled stochastic reaction-diffusion system modelling the propagation of the intracelullar and extracellular electrical potentials, and stochastic ionic currents in the heart. These equations are coupled to the stochastic quasi-static elliptic equation in the torso. Thus, we will demonstrate that the novel concept of applying the stochastic model will be useful to improve noninvasive reconstruction of electrical heart activity. We will perform numerical experiments representing the effect of the stochastic heart dynamic on the inverse solutions. Moreover, we will study the stability result for the conductivities and numerically solve the parameters estimations problem in the stochastic model.
10.1.2 Participation in other International Programs
ECOS Nord
Participants: Jacques Henry, Yves coudière.
-
Title:
Qualitative and numerical analysis of inverse problems in cardiology
-
Partner Institution(s):
Cuerpo Academico de Ecuaciones Diferenciales y Modelacion Matematica, Facultad de Ciencias Fisico Matematicas
- Benemerita Universidad Autonoma de Puebla, Mexico
-
Date/Duration:
2021-2025 (prolonged due to Covid)
- Additionnal info/keywords:
ECOS Sud
Participants: Mostafa Bendahmane, Yves coudière.
-
Title:
Virtual Element Methods for Different Bidomain Models for Cardiac Electrphysiology
-
Partner Institution(s):
Departamento de Matemática, Facultad de Ciencias
- Universidad del Bio-Bio, Conception, Chile
-
Date/Duration:
2021-2024
- Additionnal info/keywords:
10.2 International research visitors
10.2.1 Visits of international scientists
Other international visits to the team
Narjess Ben Abid
-
Status
Intern
-
Institution of origin:
ENIT TUNIS
-
Country:
Tunisia
-
Dates:
Nov. 2023
- Context of the visit:
-
Mobility program/type of mobility:
Internship
Emna Bouaine
-
Status
Intern
-
Institution of origin:
ENIT TUNIS
-
Country:
Tunisia
-
Dates:
May 2023 to Oct 2023
- Context of the visit:
-
Mobility program/type of mobility:
Internship
Hamza Ammar
-
Status
PhD
-
Institution of origin:
University of Tunis El Manar
-
Country:
Tunisia
-
Dates:
May 2023 until Jun 2023
-
Context of the visit:
SPICY associated team
-
Mobility program/type of mobility:
research stay
Moises Soto Bajo
-
Status
Researcher
-
Institution of origin:
Benemerita Universidad Autonoma de Puebla
-
Country:
Mexico
-
Dates:
June 2023 (2 weeks)
-
Context of the visit:
ECOS Nord project
-
Mobility program/type of mobility:
research stay
Andres Fraguela
-
Status
Researcher
-
Institution of origin:
Benemerita Universidad Autonoma de Puebla
-
Country:
Mexico
-
Dates:
September 2023 (1 week)
-
Context of the visit:
ECOS Nord project
-
Mobility program/type of mobility:
research stay
David Mora
-
Status
Researcher
-
Institution of origin:
Universidad del Bio Bio
-
Country:
Chile
-
Dates:
2023 (1 week)
-
Context of the visit:
ECOS Sud project
-
Mobility program/type of mobility:
research stay
Veronica Anaya
-
Status
Researcher
-
Institution of origin:
Universidad del Bio Bio
-
Country:
Chile
-
Dates:
2023 (1 week)
-
Context of the visit:
ECOS Sud project
-
Mobility program/type of mobility:
research stay
10.2.2 Visits to international teams
Research stays abroad
EmmaLagracie
-
Visited institution:
Université d'Ottawa
-
Country:
Canada
-
Dates:
From August 11th to December 3rd 2023
-
Context of the visit:
Mitacs exchange
-
Mobility program/type of mobility:
Research stay under surpervision of Prof. Yves Bourgault
persNarimaneGassa
-
Visited institution:
Simula Laboratory
-
Country:
Norway
-
Dates:
June 26th to August 18th, 2023
-
Context of the visit:
PersonalizeAF
-
Mobility program/type of mobility:
Research stay
10.3 European initiatives
10.3.1 H2020 projects
MICROCARD
Participants: Yves Coudière, Mark Potse, Zeina Chehade, Wissam Bouymedj, Laetitia Mottet, Corentin Prigent.
MICROCARD project on cordis.europa.eu
-
Title:
Numerical modeling of cardiac electrophysiology at the cellular scale
-
Duration:
From April 1, 2021 to September 30, 2024
-
Partners:
- The Inria centers in Bordeaux and Nancy (as linked third party)
- Megware GmbH, Germany
- SIMULA Research Laboratory AS, Norway
- Université de Strasbourg (UNISTRA), France
- Zuse Institute Berlin, Germany
- Università della Svizzera italiana (USI), Switzerland
- Karlsruhe Institute of Technology (KIT), Germany
- Université de Bordeaux (UBx), France
- Università degli studi di Pavia (UNIPV), Italy
- Institut Polytechnique de Bordeaux (Bordeaux INP), France
- Numericor GmbH, Austria
- Orobix SRL (OROBIX), Italy
-
Inria contact:
Mark Potse
-
Coordinator:
Mark Potse
-
Summary:
Cardiovascular diseases are the most frequent cause of death worldwide and half of these deaths are due to cardiac arrhythmia, a disorder of the heart's electrical synchronization system. Numerical models of this complex system are highly sophisticated and widely used, but to match observations in aging and diseased hearts they need to move from a continuum approach to a representation of individual cells and their interconnections. This implies a different, harder numerical problem and a 10,000-fold increase in problem size. Exascale computers will be needed to run such models.
We propose to develop an exascale application platform for cardiac electrophysiology simulations that is usable for cell-by-cell simulations. The platform will be co-designed by HPC experts, numerical scientists, biomedical engineers, and biomedical scientists, from academia and industry. We will develop, in concert, numerical schemes suitable for exascale parallelism, problem-tailored linear-system solvers and preconditioners, and a compiler to translate high-level model descriptions into optimized, energy-efficient system code for heterogeneous computing systems. The code will be parallelized with a recently developed runtime system that is resilient to hardware failures and will use an energy-aware task placement strategy.
The platform will be applied in real-life use cases with high impact in the biomedical domain and will showcase HPC in this area where it is painfully underused. It will be made accessible for a wide range of users both as code and through a web interface.
We will further employ our HPC and biomedical expertise to accelerate the development of parallel segmentation and (re)meshing software, necessary to create the extremely large and complex meshes needed from available large volumes of microscopy data.
The platform will be adaptable to similar biological systems such as nerves, and components of the platform will be reusable in a wide range of applications.
SimCardioTest
Participants: Yves Coudière, Michael Leguèbe, Gwladys Ravon, Valentin Pannetier, Arthur Jean.
SimCardioTest project on cordis.europa.eu
-
Title:
In silico testing and certification of healthcare products
-
Duration:
From January 1, 2021 to December 30, 2024
-
Partners:
- Université de Bordeaux, France
- Universidad Pompeu Fabra, Spain
- Universitat Politecnica de Valencia, Spain
- SIMULA Research Laboratory AS, Norway
- INSILICOTRIALS Technologies S.P.A., Italy
- SORIN CRM SAS, France
- EXACTCURE, France
- Boston Scientific SCIMED INC, USA
- Virtual Physiological Human Institute for Integrative Biomedical Research VZW, Belgium
-
Inria contact:
Yves Coudière
-
Coordinator:
Maxime Sermesant
-
Summary:
Computer modelling and simulation have the power to increase speed and reduce costs in most product development pipelines. The EU-funded SimCardioTest project aims to implement computer modelling, simulation and artificial intelligence to design and test cardiac drugs and medical devices. Scientists will establish a platform for running in silico trials and obtaining scientific evidence based on controlled investigations. The simulation of disease conditions and cohort characteristics has the potential to overcome clinical trial limitations, such as under-representation of groups. It also reduces the size and duration of human clinical trials as well as animal testing, and offers robust, personalised information. Leveraging in silico technology in healthcare will expedite product and drug certification and offer patients the best possible care.
Personalized AF
Participants: Nejib Zemzemi, Yves Coudière, Narimane Gassa.
PersonalizeAF project on cordis.europa.eu
-
Title:
Personalized Therapies for Atrial Fibrillation. A Translational Approach
-
Duration:
From 1st February 2020 to 31st January 2024
-
Partners:
- Karlsruhe Institute of Technology, Germany
- Universiteit Maastricht, Netherlands
- Universitätsklinikum Freiburg, Germany
- Université de Bordeaux, France
- The University of Oxford, United Kingdom
- CONSORCI Institut D'investigacions Biomediques AUGUST PI I SUNYER, Spain
- ALMA MATER STUDIORUM - UNIVERSITA DI BOLOGNA, Italy
- SIMULA Research Laboratory AS, Norway
- Fundacion para la Investigacion Biomédica del Hospital Gregorio Maranon, Spain
- NCARDIA Services BV, Netherlands
- Fundacion para la Investigacion del Hoispital Universitario La Fe de la Comunidad Valenciana, Spain
-
Inria contact:
Nejib Zemzemi
- Coordinator:
-
Summary:
Atrial Fibrillation (AF) is the most common cardiac arrhythmia affecting more than 6 million Europeans with a cost exceeding 1
PersonalizeAF addresses this challenge by delivering an innovative multinational, multi-sectorial, and multidisciplinary research and training programme in new technologies and novel strategies for individualized characterization of AF substrate to and increase treatments efficiency.
From the research point of view, PersonalizeAF will integrate data and knowledge from in-vitro, in silico, ex vivo and in vivo animal and human models to: 1) generate an individual description of the state of the atrial muscle identifying the disease mechanisms and characteristics; 2) understand the potential effect that different therapies have on different atrial substrates; and 3) combine this information to generate a specific profile of the patient and the best therapy for each patient.
With this purpose, PersonalizeAF partnership aggregates relevant scientific staff from the academic and clinical world with highly specialised biomedical companies which will be involved in a high-level personalised training programme that will train a new generation of highly skilled professionals and guarantee ESRs and future PhD students outstanding Career Opportunities in the biomedical engineering, cardiology services and medical devices sectors. PersonalizeAF will disseminate results to a wide spectrum of stakeholders, create awareness in the general public about atrial fibrillation and encourage vocational careers among young students.
10.4 National initiatives
ANR MAESTRO
The ANR project MAESTRO (Magnetic Signal detection of ventricular arrhythmOgenic substrates), coordinated by Prof. Michel Haïssaguerre (IHU Liryc), has a computational component for which we recruited a postdoc from December 2022 to mid-November 2023, directed by Mark Potse.
GENCI
GENCI project A0130307379, Interaction between tissue structure and ion-channel function in cardiac arrhythmia, coordinated by Mark Potse, comprises 2.35 million core-hours on the national supercomputers Zay and Joliot-Curie. Compared to previous years it is a modest allocation. This is because most of our computational needs in 2023 are either smaller or larger than the national scale.
Dielectric project
The Dielectric project, co-funded by the Federation Française de Cardiologie and Inria, started in January 2023, and is co-piloted by Pr. Pierre Jaïs (IHU Liryc) and Clair Poignard (Inria MONC). It aims at a better understanding of cardiac ablation by electroporation. Both Inria teams MONC and CARMEN are involved, with the PhD of Simon Bihoreau, co-directed by Annabelle Collin (MONC) and Michael Leguèbe (CARMEN).
ANR Mire4VTach
PI Annabelle Collin (Inria MONC), started late 2023. Michael Leguèbe contributes in Mire4VTach, another project on cardiac electroporation which is more focused on the application and confrontation with data than the work in the Dielectric project. Mire4Tach also involves people from Inria MONC, CARMEN and IHU Liryc.
10.5 Regional initiatives
EITCardio
Participants: Laura Bear, Yves Coudière, Charles Pierre, Bénédicte Puig, Lisl Weynans.
-
Title:
EITCardio
-
Duration:
From September 2023 to September 2027
-
Partners:
- Inria Bordeaux Sud-Ouest
- IHU-liryc
- Université de Pau et des Pays de l'Adour
-
Inria contact:
Lisl Weynans
-
Coordinator:
Lisl Weynans
-
Summary:
The objective of this project is to develop mathematical methods for solving Electrical Impedance Tomography (EIT) to enhance the resolution of the ECGi (Electrocardiographic Imaging) problem and validate them experimentally. Specifically, the project consists of two parts:
Development of mathematical and numerical methods to solve the inverse problem of EIT in the torso and identify influential parameters for the propagation of the electric field, such as conductivities and organ movements.
Experimental validation of the ECGi + EIT coupling. This experimental validation will be conducted first within the experimental setup, the torso tank, currently available at Liryc, which allows measurements for ECGi in a controlled environment. Subsequently, it will be conducted as in-vivo experiments, meaning a context closer to clinical reality.
ATLAS-RVA
Participants: Peter Langfied, Karim Benali.
-
Title:
Atlas of normal and pathological ventricular Repolarization patterns and correlation with clinical re-entrant Ventricular Arrhythmias
-
Duration:
From January 2024 to December 2025
-
Partners:
- Inria Bordeaux Sud-Ouest
- IHU-liryc
-
Inria contact:
Peter Langfield
-
Coordinator:
Peter Langfield
-
Summary:
The project aims to study the variability of patterns of ventricular repolarization times in healty and unhealthy patients, and explore the role of such patterns.
The main objective of the project is to perform a statistical assessment of patterns of ventricular repolarization time via unipolar electrogram annotations from CARTO datasets, from roughly two hundred datasets from data centers across France. The cruicial difference between this project and previous work, is that specific filtering settings were imposed for the data acquisition that make the annotations reliable. The project will exploit this particular setting and the large volumes of data, to exlpore and assess alternative methods of repolarization time annotation. Finally, the project will study cases of ventricular tachycardia with the aim of ellucidating the role of repolarization time patterns and also the associated resitituion properties of the tissue.
Peter Langfield Nejib Zemzemi Yves Coudière Corentin Prigent Laetitia Mottet Mark Potse Michael Leguebe Lisl Weynans Mostafa Bendahmane Emma Lagracie Niami Nasr Valentin Pannetier Zeina Chehade
11 Dissemination
11.1 Promoting scientific activities
11.1.1 Scientific events: organisation
General chair, scientific chair
- Yves Coudière: chair of the organisation of the General Assembly of the SimCardioTest project, March, 20-21, 2023
- Yves Coudière: organisation of the first InnovaHeart day, Bordeaux, March, 22nd, 2023, see video
Member of the organizing committees
- Peter Langfield: Co-organizer of mini-symposium at EQUADIFF 2024 conference.
11.1.2 Scientific events: selection
Member of the conference program committees
- Yves Coudière, Nejib Zemzemi: Program Committee for the FIMH 2023 international conference
- Yves Coudière: program Committe for the first LiryConnect days, November 14-15, 2023
11.1.3 Invited talks
- Yves Coudière, invited talk at RICAM Workshop 2 on Cardiovascular Modeling and Simulation, 2023-11-13 to 2023-11-17, Linz, Austria.
- Yves Coudière, invited talk at the JPVV66 workshop in honor of J.-P. Vila, 2023-04-04 to 2023-04-06, Banyuls, France.
- Mark Potse. MICROCARD Overview. openCARP User Meeting, Karlsruhe, Germany, May 2023.
- Corentin Prigent and Laetitia Mottet. Meshing with the Mmg platform. openCARP User Meeting, Karlsruhe, Germany, May 2023.
- Laetitia Mottet. Challenges in meshing tissue at the micro-scale for cardiac modeling. IHU Liryc International Scientific Workshop, Pessac, France, November 2023.
- Mark Potse. Large Numerical modeling of cardiac electrophysiology at the cellular scale. EuroHPC Summit: European Supercomputing Excellence in the Exascale Era, Göteborg, Sweden, March 2023.
- Mark Potse. Digital Twins in Cardiology. Inserm/Inria Digital Twins for Health seminar, Brussels, 28 March 2023.
- Mark Potse. Simulating Cardiac Electrophysiology: The Quest for Detail and Diversity. Keynote talk at the Measurement Science congress, Smolenice, Chech republic, 29 May 2023.
- Mark Potse. Numerical modeling of cardiac electrophysiology at the cellular scale. IHU Liryc International Scientific Workshop, November 2023.
- Mark Potse. Numerical modeling of cardiac electrophysiology at the cellular scale. Symposium “Where computation meets cardiology”, Maastricht, The Netherlands, November 2023.
- Lisl Weynans, seminar of laboratory MIP, Toulouse
- Lisl Weynans, seminar of laboratory LMAP, Pau
11.1.4 Leadership within the scientific community
Mark Potse is leading the H2020-EuroHPC MICROCARD project, strengthening his role as a European leader in the scientific community.
11.1.5 Scientific expertise
- Peter Langfield: Project reviewer for a proposal at Heart Research UK (HRUK).
- Yves Coudière: diverse expertises, including applications to IHU Liryc internal call, CIFRE applications, Inserm projects.
- Mark Potse: associate editor for Frontiers in Cardiac Electrophysiology, reviewer for several journals and congresses.
11.1.6 Research administration
- Comité de direction IHU Liryc: Yves Coudière participates to these bi-monthly commmittees
- Conseil scientifique IHU Liryc: Yves Coudière, Michael Leguèbe, Mark Potse participate to the scientific committe of IHU Liryc (a few meetings in 2023)
- Mark Potse is a member of the IDRIS user committee
- Lisl Weynans is a member of the BCP of Bordeaux Sud Ouest Inria center.
11.2 Teaching - Supervision - Juries
11.2.1 Teaching
The 2 assistant professors and 1 professor of the team teach at several levels of the Bordeaux University programs in Mathematics, Neurosciences, and Medicine (respectively, 192, 192 and 96 h/year on average). The researchers also have a regular teaching activity, contributing to several courses in the Applied Mathematics at the Bachelor and Master levels (between 16 and 72 h/year).
The PhD students who ask for it are used to teach between 32 and 64 h/year, usually courses of general mathematics in L1 or mathematics for biologists in L1 or L2.
Typical courses taught by team members (L for Bachelor level, M for Master level):
- Numerical analysis (L2)
- Programming for scientific computing with C++ (L3) and Modern Fortran (M1)
- Solving sparse linear systems (L3)
- Numerical approximation of PDEs: Finite Differences, Finite Elements, Finite Volumes (M1, M2)
- Supervision of programming projects (L3, M1)
- Linear Algebra, Optimization under constraints (L2 and Essca school)
- Analysis, L2
- Computational Neurosciences, M2
- Neuropsychology and Psychophysiology, L3
- Physics for students in medicine, one lecture on cardiac modelling
11.2.2 Juries
- Yves Coudière: member of the jury for the PhD of N. Nasr (University of Bordeaux), reviewer and member of the jury for the PhD of L. Gander (Universita della Svizzera Italiana)
- Lisl Weynans: member of the jury for the PhD of N. Nasr
- Mark Potse: co-advisor of Hassaan Bukhari (PhD defense in February 2023)
- Lisl Weynans: member of the jury for the PhD of T. Carlier (Bordeaux), for the PhD of Z.Yang (Paris-Saclay)
- Lisl Weynans: reviewer of the PhD manuscript of C. Chambon (CNAM Paris), of the PhD manuscript of L. Salomon (Ensta Bretagne)
11.2.3 Teaching administration
- Yves Coudière: elected member of the conseil du département des Sciences et Technologies of Bordeaux University. The committe votes on all matters related to teaching at the University in the domain of sciences and technologies.
- Yves Coudière: elected member of the conseil de l'Unité de Formation Mathématiques et Interaction. This committee discusses and votes on all matters related to teaching in the department of mathematics, providing helpful decisions to the above committee.
- Lisl Weynans: responsible for the Licence de Mathématique parcours ingénierie mathématique (i.e. Bachelor on applied mathematics),
- Lisl Weynans: responsible for a course (called mineure Mathématiques du parcours International de la Licence,
- Mostafa Bendahmane: responsible for international mobility of students from the Licence MIASHS, which is the Bachelor on mathmatics applied to social and human sciences.
11.3 Popularization
11.3.1 Articles and contents
- Yves Coudière: radio interview for Radio Campus at the Nuit Européenne de Chercheurs 2023.
- Videos on digital twins for cardiology from the InnovaHeart day: main video, Episode 1: Why create a digital heart twin?, Episode 2 : How are you contributing ?, 3- What motivates your work on the heart's digital twin?, 4: What are the remaining challenges?.
11.3.2 Education
- Whole team: participation to welcoming high school pupils for short internships.
- Laetitia Mottet: CHICHE at Lycée (high school) Jay de Beaufort, Périgueux, December 2023.
- Yves Coudière: CHICHE at 3 Lycées (high school), January, February, November 2023.
- Yves Coudière: small workshop for high school pupils during the Fête de la Science 2023.
11.3.3 Interventions
- Moi Mathématicienne, Moi Informaticienne (MIMM) : organisation (L. Weynans) and participation (L. Weynans, E. Lagracie) to a week organized during school holidays to welcome young women high-school students at the University.
- During the GA of SimCardiotest, on March, 21, we organized a small exchange between cardiologists, representatives of patients' organizations, and scientists to discuss on in-silico trial and related issues in science and the society.
12 Scientific production
12.1 Major publications
- 1 articleConvergence of discrete duality finite volume schemes for the cardiac bidomain model.Networks and Heterogeneous Media622011, 195-240HALback to text
- 2 articleA mathematical model of Purkinje-Muscle Junctions.Mathematical Biosciences and Engineering842011, 915-930
- 3 articleExistence And Uniqueness Of The Solution For The Bidomain Model Used In Cardiac Electrophysiology.Nonlinear Anal. Real World Appl.1012009, 458-482URL: http://hal.archives-ouvertes.fr/hal-00101458/fr
- 4 articleA 2D/3D Discrete Duality Finite Volume Scheme. Application to ECG simulation.International Journal on Finite Volumes612009, URL: http://hal.archives-ouvertes.fr/hal-00328251/frback to text
- 5 articleStability And Convergence Of A Finite Volume Method For Two Systems Of Reaction-Diffusion Equations In Electro-Cardiology.Nonlinear Anal. Real World Appl.742006, 916--935URL: http://hal.archives-ouvertes.fr/hal-00016816/frback to text
- 6 articleThe Early Repolarization Pattern; A Consensus Paper.Journal of the American College of Cardiology66resulting from the Symposium on J Wave Patterns and a J Wave Syndrome, Glasgow, August 2013. Defines terminology for the J point: Jo, Jp, Jt. Provisionally accepted 19 May 2015.2015, 470-477URL: http://dx.doi.org/10.1016/j.jacc.2015.05.033back to text
- 7 articleReduced Sodium Current in the Lateral Ventricular Wall Induces Inferolateral J-Waves.Front Physiol7365August 2016HALDOIback to textback to text
- 8 articlePreconditioning the bidomain model with almost linear complexity.Journal of Computational Physics2311January 2012, 82--97URL: http://www.sciencedirect.com/science/article/pii/S0021999111005122DOIback to text
12.2 Publications of the year
International journals
International peer-reviewed conferences
Conferences without proceedings
Doctoral dissertations and habilitation theses
Reports & preprints
Other scientific publications
12.3 Cited publications
- 31 inproceedingsAn Improved Iterative Pace-Mapping Algorithm to Detect the Origin of Premature Ventricular Contractions.Computing in Cardiology47abstract nr 62, really early. Oral presentation.RiminiComputing in Cardiology2020, 62HALDOIback to text
- 32 bookAssessing the credibility of computational modeling through verification and validation: Application to medical devices.2018back to text
- 33 inproceedingsModélisation et simulation de l'électrophysiologie cardiaque à l'échelle microscopique.43e Congrès National d'Analyse Numérique (CANUM)oral presentationSMAIObernai, Alsace, FranceMay 2016, URL: http://smai.emath.fr/canum2016/resumesPDF/peb/Abstract.pdfback to textback to text
- 34 miscTheoretical and Numerical Study of Cardiac Electrophysiology Problems at the Microscopic Scale..PosterJuly 2016HALback to text
- 35 inproceedingsA Three-Dimensional Computational Model of Action Potential Propagation Through a Network of Individual Cells.Computing in Cardiology 2017Rennes, FranceSeptember 2017, 1-4HALback to text
- 36 inproceedingsMicroscopic Simulation of the Cardiac Electrophysiology: A Study of the Influence of Different Gap Junctions Models.Computing in CardiologyMaastricht, NetherlandsSeptember 2018HALback to text
- 37 articleImpact of Septal Radiofrequency Ventricular Tachycardia Ablation; Insights From Magnetic Resonance Imaging.Circulation1302014, 716-718URL: https://www.ahajournals.org/doi/10.1161/CIRCULATIONAHA.114.010175back to text
- 38 inproceedingsDo we need to enforce the homogeneous Neuman condition on the Torso for solving the inverse electrocardiographic problem by using the method of fundamental solution ?Computing in Cardiology 201643Computing in Cardiology 2016Vancouver, CanadaSeptember 2016, 425-428HALback to text
- 39 articleOptimal monodomain approximations of the bidomain equations used in cardiac electrophysiology.Mathematical Models and Methods in Applied Sciences246February 2014, 1115-1140HALback to textback to text
- 40 articleSpatially Coherent Activation Maps for Electrocardiographic Imaging.IEEE Transactions on Biomedical Engineering64May 2017, 1149-1156HALDOIback to text
- 41 inproceedingsEpicardial Fibrosis Explains Increased Transmural Conduction in a Computer Model of Atrial Fibrillation .Computing in CardiologyVancouver, CanadaSeptember 2016HALback to textback to textback to text
- 42 articleSudden cardiac arrest associated with early repolarization.N. Engl. J. Med.3582008, 2016--2023back to text
- 43 articleSpontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins.N. Engl. J. Med.3391998, 659-666URL: https://www.nejm.org/doi/full/10.1056/NEJM199809033391003back to text
- 44 articleMechanism of Right Precordial ST-Segment Elevation in Structural Heart Disease: Excitation Failure by Current-to-Load Mismatch.Heart Rhythm72010, 238-248URL: http://dx.doi.org/10.1016/j.hrthm.2009.10.007back to text
- 45 articleA microstructural model of reentry arising from focal breakthrough at sites of source-load mismatch in a central region of slow conduction.Am. J. Physiol. Heart Circ. Physiol.3062014, H1341-1352back to text
- 46 inproceedingsA new ECG-based method to guide catheter ablation of ventricular tachycardia.iMAging and eLectrical TechnologiesUppsala, SwedenApril 2018HALback to text
- 47 inproceedingsA practical algorithm to build geometric models of cardiac muscle structure.ECCOMAS 2022 - The 8th European Congress on Computational Methods in Applied Sciences and EngineeringOslo, NorwayJune 2022HALback to text
- 48 articleA Comparison of monodomain and bidomain reaction-diffusion.IEEE Transactions on Biomedical Engineering53122006, 2425-2435URL: http://dx.doi.org/10.1109/TBME.2006.880875back to text
- 49 articleCardiac Anisotropy in Boundary-Element Models for the Electrocardiogram.Medical and Biological Engineering and Computing472009, 719--729URL: http://dx.doi.org/10.1007/s11517-009-0472-xback to text
- 50 inproceedingsAnatomically-induced Fibrillation in a 3D model of the Human Atria.Computing in CardiologyMaastricht, NetherlandsSeptember 2018HALback to text
- 51 miscRegional conduction slowing can explain inferolateral J waves and their attenuation by sodium channel blockers.PosterSeptember 2016HALback to text
- 52 inproceedingsFeasibility of Whole-Heart Electrophysiological Models With Near-Cellular Resolution.CinC 2020 - Computing in CardiologyRimini / Virtual, ItalySeptember 2020HALDOIback to text
- 53 articleScalable and Accurate ECG Simulation for Reaction-Diffusion Models of the Human Heart.Frontiers in Physiology9April 2018, 370HALDOIback to text
- 54 articleA Cell-Based Framework for Numerical Modeling of Electrical Conduction in Cardiac Tissue.Front. Phys.5This is very very similar to what we were doing with PEB... Looks like we're scooped at least for the approach, but we do have a few abstracts ([becue:cinc17], [becue16a], MMCE meeting in Ottawa November 2017). Note it's in Frontiers in (Biomedical) Physics, not Physiology.2017, 48back to textback to text
